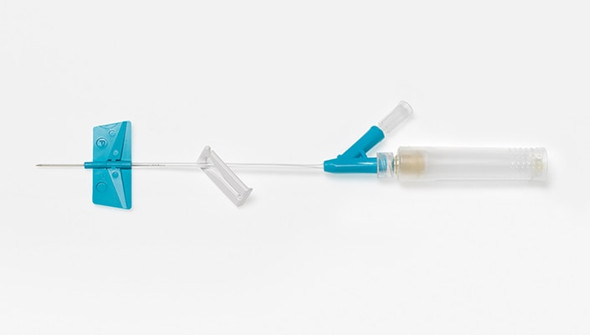
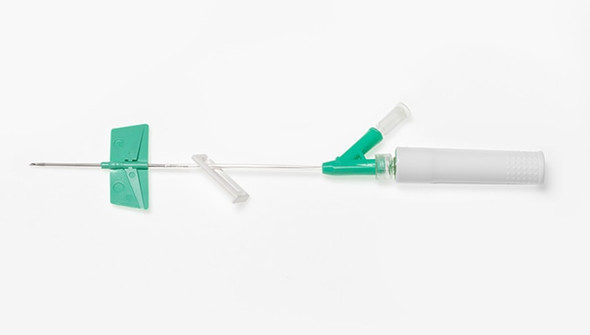
Description
BD Saf-T-Intima Safety System with BD Vialon Catheter Material is designed for clinicians who need reliable vascular access with enhanced safety. This closed IV catheter system supports efficient fluid delivery and helps reduce blood exposure during insertion. Ideal for hospital and clinical environments where minimizing risk and maintaining workflow are priorities, thanks to its integrated blood control feature.
- Blood Control Technology – Minimizes blood exposure during insertion, supporting a cleaner and safer procedure.
- BD Vialon Catheter Material – Offers flexibility and durability, helping users feel confident in every application.
- High Flow Rate – Delivers fluids efficiently at 22 mL/min, ensuring timely patient care and peace of mind.
- Y Adapter Included – Facilitates multiple access points, making setup smoother for clinicians.
- Color-Coded Hub – Yellow hub allows quick gauge identification, reducing errors and saving time.
Closed IV Catheter System for Reliable Vascular Access
This closed IV catheter system is engineered for consistent performance in demanding healthcare settings. The BD Saf-T-Intima Safety System features a 24 G (0.7 mm) catheter with a 0.75 in (19 mm) length, supporting precise placement and patient comfort. Its integrated blood control mechanism helps reduce the risk of blood exposure, while the BD Vialon catheter material enhances flexibility and durability. With a high flow rate of 22 mL/min and a pressure rating of 45 psi, this device is suitable for a range of infusion needs. Clinicians in hospitals and outpatient clinics benefit from the color-coded yellow hub, which streamlines gauge selection and workflow. Designed for those who prioritize safety and efficiency, this vascular access device delivers steady results and supports smoother procedures.
Features and Benefits
Safety and Efficiency
- Blood Control Feature – Reduces risk of blood exposure, supporting a safer clinical environment.
- Closed IV System – Helps maintain a sterile pathway, minimizing contamination risk for patients and staff.
Performance and Flow
- 22 mL/min Gravity Flow Rate – Enables rapid fluid administration for urgent care scenarios.
- High Pressure Rating – Withstands up to 45 psi, suitable for various infusion protocols.
Material and Design
- BD Vialon Catheter Material – Provides flexibility and strength for reliable vascular access.
- Color-Coded Yellow Hub – Allows quick identification of gauge size, reducing setup time.
- Y Adapter – Offers multiple access options for versatile clinical use.
Product Specifications
Dimensions and Capacity
- Catheter Gauge Size: 24 G (0.7 mm)
- Catheter Length: 0.75 in (19 mm)
- Catheter Inner Diameter: 0.52 mm
- Catheter Outer Diameter: 0.7 mm
- Gravity Flow Rate: 22 mL/min
- High Pressure Rating: 45 psi
Materials and Identification
- Catheter Material: BD Vialon
- Hub Color: Yellow
- Sterilization Method: EtO
Packaging and Shelf Life
- Quantity Per Box: 25
- Quantity Per Case: 200
- Total Shelf Life: 4 years
Explore More: See all Peripheral IV Catheters | Shop more BD Saf-T-Intima Products
Enhanced Safety and Workflow for Clinical Teams
Choose this vascular access device for its focus on safety, efficiency, and ease of use. The integrated blood control feature and closed system design help reduce contamination risks, while the color-coded hub and Y adapter streamline clinical workflow. This product is built for clinicians who value reliable performance and patient comfort in every procedure.
- Integrated Blood Control – Minimizes exposure risk, supporting a safer work environment for staff.
- Closed System Design – Helps maintain sterility, giving clinicians confidence during use.
- Quick Gauge Identification – Yellow hub speeds up setup, reducing the chance of errors.
- Versatile Y Adapter – Allows multiple access points, making procedures more efficient.
- Proven Material Durability – BD Vialon construction stands up to repeated use, offering peace of mind.
Frequently Asked Questions (FAQs)
What is the gauge and length of this catheter?
This catheter is 24 G (0.7 mm) in diameter and 0.75 in (19 mm) long, supporting precise vascular access.
How does the blood control feature work?
The integrated blood control mechanism helps minimize blood exposure during insertion, supporting safer procedures for clinicians.
What is the flow rate for this device?
The gravity flow rate is 22 mL/min, allowing for efficient fluid administration in clinical settings.
What material is used for the catheter?
The catheter is made from BD Vialon material, which provides flexibility and durability for reliable use.
How is the device sterilized?
This product is sterilized using EtO (ethylene oxide) to ensure safety and cleanliness.
What is the shelf life of this product?
The total shelf life is 4 years, supporting long-term storage and readiness.
How is the product packaged?
Each box contains 25 units, and each case contains 200 units for efficient inventory management.