Description
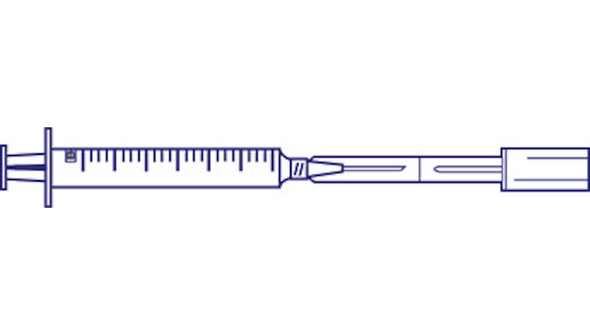
BD Twinpak Dual Cannula Device with 3 Ml Syringe is designed for clinicians who need a reliable, sterile solution for syringe filling and needleless IV fluid delivery. This device supports safe, efficient workflows in hospital and clinical settings, especially when working with preslit injection sites and compatible needleless systems.
- Dual Cannula Design – Enables both syringe filling and needleless IV fluid delivery, supporting streamlined procedures.
- Content Sterile – Ensures a high standard of safety for patient care, giving you peace of mind.
- Opaque Red Hub – Provides clear visual identification, reducing the risk of connection errors.
- Bulk Packaging – 800 units per case for efficient stocking and supply management.
Needleless Access Device for Safe and Efficient Fluid Delivery
This dual cannula device is specifically designed for use with preslit injection sites and vials intended for needleless access. It is not compatible with conventional injection sites that are not preslit. The system includes an opaque red hub for easy identification and a blunt plastic cannula for safe IV fluid delivery or aspiration. Clinicians in hospitals and infusion centers benefit from streamlined workflows and reduced risk of needlestick injuries. The sterile packaging and bulk case count help maintain readiness for high-volume environments. With its compatibility for Abbott LifeShield, McGaw SafeLine, and Baxter InterLink systems, this device supports a wide range of clinical needs while prioritizing safety and efficiency.
Features and Benefits
Safe and Efficient Fluid Handling
- Blunt Plastic Cannula – Designed for use with preslit injection sites, reducing the risk of accidental needlestick.
- Needleless Access – Facilitates safe delivery and aspiration of IV fluids without conventional needles.
Visual Safety and Identification
- Opaque Red Hub – Enhances visibility for quick and accurate device recognition.
- Distinctive Design – Helps prevent connection errors in busy clinical settings.
Bulk Supply and Sterility
- Content Sterile – Maintains a sterile field for every procedure.
- Bulk Packaging – 800 units per case support high-volume clinical use.
Product Specifications
Dimensions and Weight
- Case Dimensions: 21.7 in x 15.6 in x 15.6 in
- Weight: 17.3 lb
Packaging and Quantity
- Quantity Per Case: 800
- Unit of Measure: Per Case
Sterility and Identification
- Sterility: Content sterile
- Color of Tubing: None
- Opaque Red Hub: Yes
Technical Details
- Number of Backcheck Valves: 0
- Number of Connectors: 0
- Number of Fuses: 0
- Number of Y Sites: 0
- Stopcock: None
- T Connector Type: None
- Type of Connector: None
- Y Site Port Type: None
Explore More: See all IV Accessories | Shop more BD Products
Reliable Performance for High-Volume Clinical Environments
This device stands out for its dual cannula design and sterile packaging, supporting busy healthcare teams who need dependable, ready-to-use supplies. The clear visual cues and compatibility with leading needleless systems help reduce errors and improve workflow. You can count on consistent performance and peace of mind in every procedure.
- Dual Functionality – Supports both syringe filling and needleless delivery, increasing versatility for clinicians.
- High-Volume Packaging – 800 units per case minimize restocking interruptions.
- Visual Safety Features – Opaque red hub and distinctive design help prevent misconnection.
- Designed for Compatibility – Works with Abbott LifeShield, McGaw SafeLine, and Baxter InterLink systems.
Frequently Asked Questions (FAQs)
What is the BD Twinpak Dual Cannula Device used for?
This device is used for syringe filling and needleless IV fluid delivery with preslit injection sites and compatible vials.
Is the device sterile?
Yes, the device is content sterile to help maintain safety during clinical procedures.
How many units are included per case?
Each case contains 800 units, making it suitable for high-volume environments.
What systems is this device compatible with?
It is intended for use with Abbott LifeShield, McGaw SafeLine, and Baxter InterLink systems.
Can this device be used with conventional injection sites?
No, it is designed specifically for preslit injection sites and is not compatible with conventional sites.
What is the color of the hub?
The hub is opaque red for easy identification and safety.
Does the device contain any backcheck valves or connectors?
No, this device does not include backcheck valves or connectors.