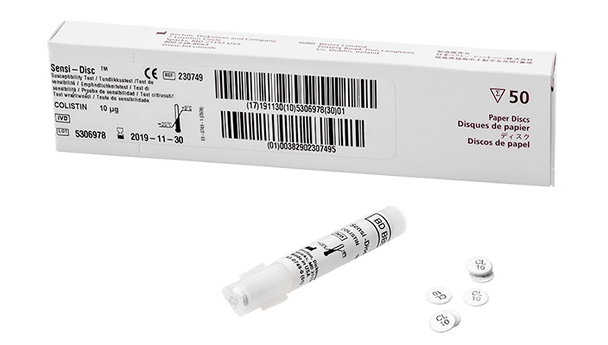
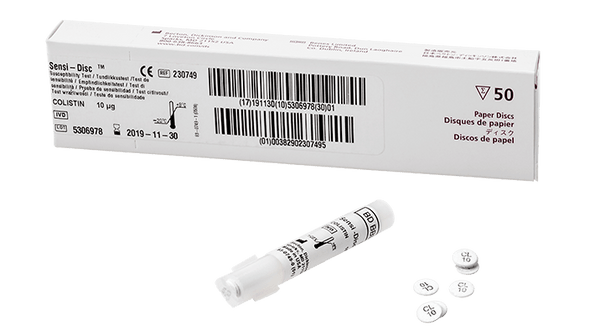
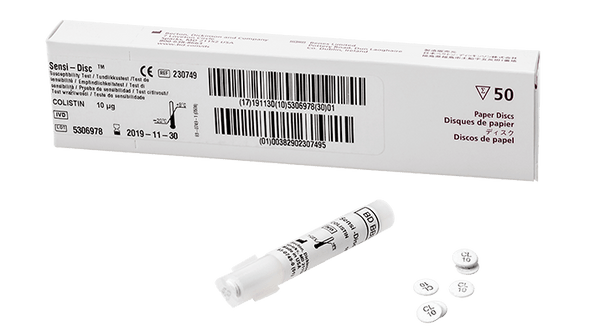
Description
BD BBL Sensi-Disc Sulfamethoxazole/trimethoprim SXT-23.75/1.25 supports laboratories seeking reliable antimicrobial susceptibility testing. Designed for clinical microbiology professionals, this product helps ensure accurate results and efficient workflows. Ideal for labs that require consistent disc diffusion performance, each disc delivers standardized concentrations for dependable testing outcomes.
- Standardized Antimicrobial Concentration – Ensures consistent and reproducible results, supporting confidence in daily lab operations.
- Convenient 10-Disc Pack – Simplifies inventory management, making routine testing easier for busy laboratory teams.
- Extended 1095-Day Shelf Life – Reduces waste and supports long-term storage, providing peace of mind for supply planning.
- Disc Diffusion Method – Widely recognized for antimicrobial susceptibility testing, helping labs meet clinical standards.
Reliable Antimicrobial Susceptibility Testing for Clinical Laboratories
This antimicrobial susceptibility test disc is designed to help clinical laboratories deliver accurate and timely results. The product uses the disc diffusion method, a trusted approach for determining the effectiveness of antibiotics against specific bacteria. With a convenient 10-disc pack and a long shelf life, it supports efficient workflows and reduces the need for frequent reordering. Laboratories benefit from standardized concentrations, which help maintain consistency across tests and support quality patient care. This solution is ideal for microbiology labs focused on reliable, evidence-based testing.
Features and Benefits
Consistent Performance
- Accurate Disc Formulation – Delivers precise concentrations of sulfamethoxazole and trimethoprim for dependable test results.
- Reproducible Outcomes – Supports standardized testing protocols, helping labs maintain quality and compliance.
Operational Efficiency
- Packaged for Convenience – 10 discs per pack streamline storage and daily use in laboratory settings.
- Long Shelf Life – 1095 days of shelf stability reduces the frequency of restocking and minimizes waste.
Trusted Methodology
- Disc Diffusion Technique – Recognized by clinical microbiology standards for antimicrobial susceptibility testing.
- Backed by Research – Referenced in EUCAST evaluations for quality assurance in laboratory environments.
Product Specifications
General Information
- Product Category: BD BBL disc diffusion
- Pack Count: 10 discs
- Antimicrobial Agents: Sulfamethoxazole/Trimethoprim SXT-23.75/1.25
Identification and Codes
- SKU: 231539
- GTIN (Each): 00382902315391
- GTIN (Shelfpack): 30382902315392
Storage and Shelf Life
- Shelf Life: 1095 days
Explore More: See all BD BBL Disc Diffusion | Shop more BD BBL Products
Choose Consistency and Confidence for Your Lab
Laboratories rely on accurate, efficient, and standardized solutions for antimicrobial susceptibility testing. This disc diffusion product stands out by offering a long shelf life and reliable performance, helping your team maintain confidence in every result. With trusted methodology and convenient packaging, it supports smooth daily operations and quality patient care.
- Long Shelf Life Advantage – Outlasts typical inventory cycles, reducing the risk of expired stock and supporting uninterrupted workflows.
- Standardized Testing – Ensures every disc meets strict quality benchmarks for reproducibility and accuracy.
- Convenient Pack Size – 10-disc format fits routine lab needs and simplifies supply management.
- Backed by Research – Referenced in EUCAST evaluations for laboratory quality assurance.
Frequently Asked Questions (FAQs)
What is the BD BBL Sensi-Disc Sulfamethoxazole/Trimethoprim SXT-23.75/1.25 used for?
This product is used for antimicrobial susceptibility testing in clinical microbiology laboratories using the disc diffusion method.
How many discs are included in each pack?
Each pack contains 10 antimicrobial susceptibility test discs.
What is the shelf life of these discs?
The discs have a shelf life of 1095 days, supporting long-term storage and planning.
Which antimicrobial agents are present in these discs?
Each disc contains sulfamethoxazole and trimethoprim at standardized concentrations.
Are these discs recognized by clinical standards?
Yes, the disc diffusion method is widely recognized for antimicrobial susceptibility testing in clinical laboratories.
Is this product referenced in research or evaluations?
Yes, it is referenced in EUCAST evaluations for quality assurance in laboratory testing.
What is the SKU for this product?
The SKU for this antimicrobial susceptibility test disc pack is 231539.