Description
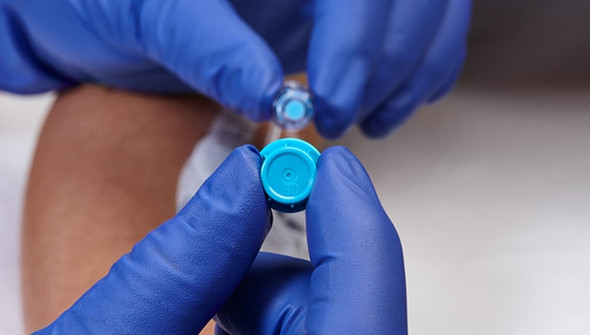
BD Site-Scrub Disinfecting Device is designed for clinicians who need reliable disinfection of injection ports and female luer hubs. When infection control is a top priority, this device delivers consistent results with every use. Its patented foam fingers and 70% isopropyl alcohol solution help support safe IV access in busy hospital and clinic environments.
- Patented Foam Fingers – Provide active friction scrubbing for thorough cleaning, giving you peace of mind during IV procedures.
- 70% Isopropyl Alcohol Solution – Ensures effective disinfection, supporting confidence in sterile technique.
- IV Pole Mount Design – Keeps the device accessible at the point of care, making daily routines easier for staff.
- Sterile, Single-Use Packaging – Reduces cross-contamination risk, helping maintain a safe environment.
Active Disinfection for Injection Ports and Luer Hubs
This disinfecting device is engineered for use on injection ports and female luer hubs, providing active friction cleaning with every application. The combination of patented foam fingers and a 70% IPA solution helps remove contaminants and supports infection control protocols. Designed for healthcare professionals in hospitals, clinics, and outpatient settings, it offers a practical solution for maintaining sterile IV access. The IV pole mount keeps the device within reach, streamlining workflow and reducing setup time. With its single-use, sterile packaging, this product helps you feel confident in your infection prevention measures.
Features and Benefits
Advanced Cleaning Technology
- Foam Finger Design – Delivers active friction scrubbing, improving cleaning effectiveness for luer surfaces.
- 70% IPA Formula – Provides rapid and reliable disinfection, supporting best practices in IV care.
Convenient and Safe Use
- IV Pole Mount – Keeps the device easily accessible for clinicians, saving time during procedures.
- Sterile, Single-Use – Minimizes contamination risk, supporting a safer clinical environment.
Product Specifications
Dimensions and Weight
- Weight: 2.3 grams
Packaging and Identification
- Quantity Per Box: 25/box
- Boxes Per Case: 40/case
- GTIN (Box): 10801741086936
- GTIN (Case): 20801741086933
- SKU: 3882025
Explore More: See all IV Catheter Disinfecting Devices | Shop more Site-Scrub Products
Reliable Disinfection for Clinical Settings
This product stands out for its patented cleaning technology and easy access design. It helps clinicians maintain high standards of infection control while streamlining daily routines. The sterile, single-use format reassures staff and patients alike that safety is always a priority.
- Patented Cleaning Mechanism – Unique foam fingers deliver consistent friction, supporting thorough disinfection.
- Accessible IV Pole Mount – Keeps supplies within arm’s reach, reducing workflow interruptions.
- Single-Use Sterility – Each device is individually packaged to lower cross-contamination risk.
- Trusted Brand – Manufactured by BD, a leader in infection prevention solutions.
Frequently Asked Questions (FAQs)
What is the BD Site-Scrub Disinfecting Device used for?
This device is intended for disinfecting injection ports and female luer hubs in clinical settings.
What is the active ingredient in the device?
The device contains 70% isopropyl alcohol (IPA) for effective disinfection.
Can the device be left on the injection port after use?
No, it should not be left on the port or hub after disinfecting. Remove immediately after use.
Is the device single-use?
Yes, each device is sterile and intended for single use only.
How is the device mounted for easy access?
It features an IV pole mount design to keep it accessible at the point of care.
What is the packaging quantity?
Each box contains 25 devices, and there are 40 boxes per case.
Are there any warnings for use?
Do not use on devices that access the nervous system. Always consult product labels for full instructions and warnings.