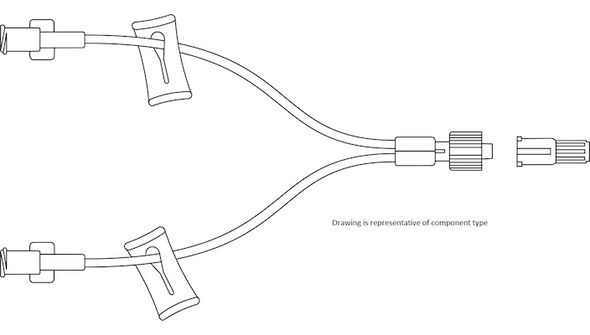
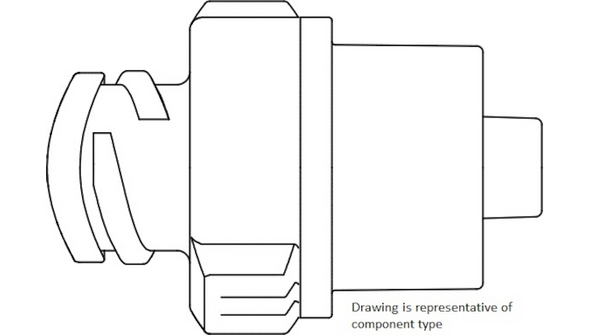
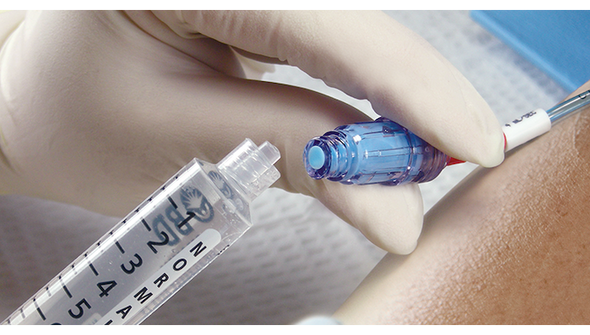
Description
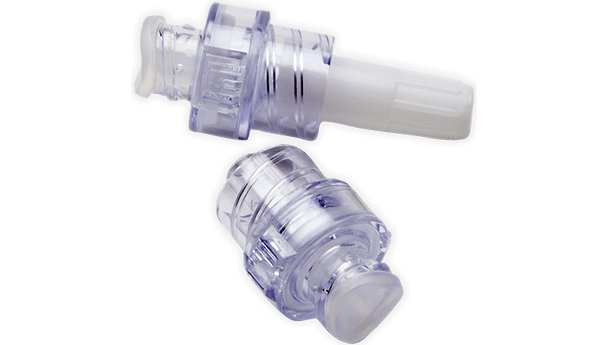
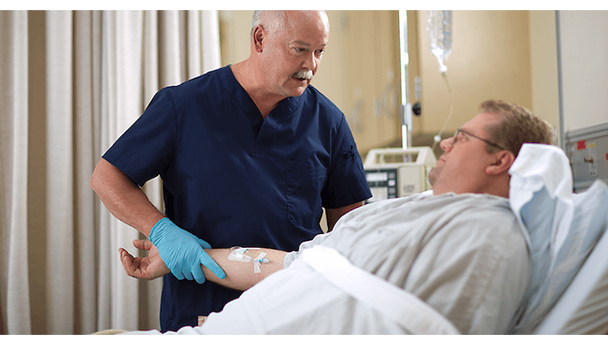
BD Q-Syte Needle-Free Connector is designed for clinicians and caregivers who need a reliable, sterile connection for intravascular fluid administration. This device helps reduce the risk of contamination and supports efficient workflows in busy healthcare settings. Its compact size and needle-free design make it ideal for hospitals, infusion centers, and outpatient care, where safety and ease of use are essential.
- Needle-Free Access – Enables safe, closed-system fluid administration, supporting infection control and peace of mind.
- Male Luer Lock Connection – Provides a secure, standardized fit for compatibility with vascular access devices, ensuring confidence in every use.
- Low Priming Volume – 0.16 mL priming volume reduces medication waste, helping you work efficiently.
- Content Sterile – Sterile packaging supports patient safety and clinical best practices, giving you reassurance during procedures.
- DEHP-Free and Latex-Free – Designed without DEHP or natural latex to minimize exposure risks and support sensitive patients.
Reliable Needle-Free Connector for Safe Fluid Administration
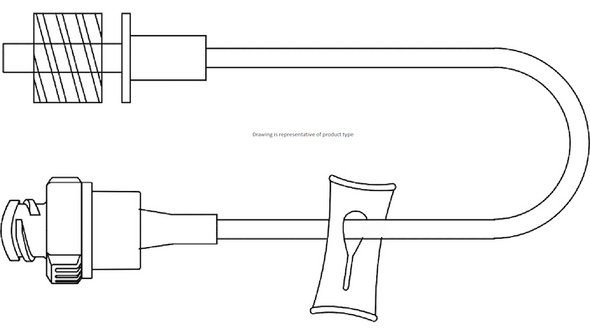
This sterile, single-use connector is engineered for use with vascular access devices in a variety of clinical environments. The needle-free design helps reduce the risk of needlestick injuries and supports closed-system protocols. With a male luer lock connection and a compact 2 cm (0.8 in) length, it fits seamlessly into standard IV setups. The low 0.16 mL priming volume minimizes medication loss, making it a practical choice for high-volume infusion settings. Its DEHP-free and latex-free construction addresses the needs of sensitive patients and aligns with modern safety standards. Designed for hospitals, clinics, and outpatient care, this connector helps streamline workflows and maintain a sterile field.
Features and Benefits
Safe and Efficient Fluid Delivery
- Needle-Free Split Septum – Supports closed-system access to reduce contamination risk and promote safety.
- Low Priming Volume – 0.16 mL volume helps conserve medication and reduces waste in every procedure.
- Content Sterile Packaging – Ensures the connector is ready for immediate clinical use, supporting infection prevention.
Compatibility and Construction
- Male Luer Lock – Standardized connection fits a wide range of vascular access devices for versatile use.
- DEHP-Free and Latex-Free – Manufactured without DEHP or natural latex to support patient safety and minimize allergic reactions.
- Compact Size – 2 cm (0.8 in) length integrates easily into IV lines without adding bulk.
Regulatory and Quality Standards
- CE Marked – Meets European regulatory standards for quality and safety.
- Country of Manufacture – Made in Mexico under strict quality controls.
Product Specifications
Dimensions and Weight
- Approximate Length: 0.8 in (2 cm)
- Weight: 1.8 lb (0.82 kg)
Technical Details
- Priming Volume: 0.16 mL
- Type of Connector: Q-Syte
- Luer Lock Type: Male Luer Lock
- Number of Connectors: 1
- Product Sub Type: Priming Vol <1mL
- Sterility: Content Sterile
- DEHP-Free: Yes
- Natural Latex: No
Regulatory and Packaging
- CE Marking: Yes
- Quantity Per Case: 200
- Case Dimensions: 10.3 in x 7.9 in x 4.8 in (26.2 cm x 20.1 cm x 12.2 cm)
Explore More: See all Needle-free Connectors | Shop more BD Q-Syte Products
Trusted Performance for Modern Clinical Settings
Choose this needle-free connector for a balance of safety, compatibility, and efficiency. Its sterile, DEHP-free, and latex-free construction helps protect both patients and staff. The compact design and low priming volume make it a practical solution for busy infusion teams. Rely on this connector to support your infection control protocols and provide peace of mind in every procedure.
- Closed-System Safety – Needle-free access reduces exposure risks, supporting a safer clinical environment.
- Efficient Medication Use – Low priming volume helps conserve costly medications and supports precise dosing.
- Universal Compatibility – Male luer lock fits most vascular access devices, simplifying inventory and setup.
- Regulatory Compliance – CE marked and manufactured to high quality standards for added confidence.
- Bulk Packaging Advantage – 200 connectors per case support high-volume facilities and reduce restocking frequency.
Frequently Asked Questions (FAQs)
What is the priming volume of this connector?
The priming volume is 0.16 mL, which helps minimize medication waste during setup.
Is this connector compatible with standard IV lines?
Yes, the male luer lock design is compatible with most vascular access devices used in clinical settings.
Does this product contain DEHP or natural latex?
No, it is manufactured without DEHP and natural latex to support patient safety and reduce allergy risks.
Is the connector supplied sterile?
Yes, each connector is supplied in content sterile packaging for immediate clinical use.
How many connectors are included per case?
Each case contains 200 connectors, suitable for high-volume healthcare environments.
Where is this product manufactured?
This connector is made in Mexico under strict quality controls.
Is this connector CE marked?
Yes, it is CE marked to meet European regulatory standards for safety and quality.