Description
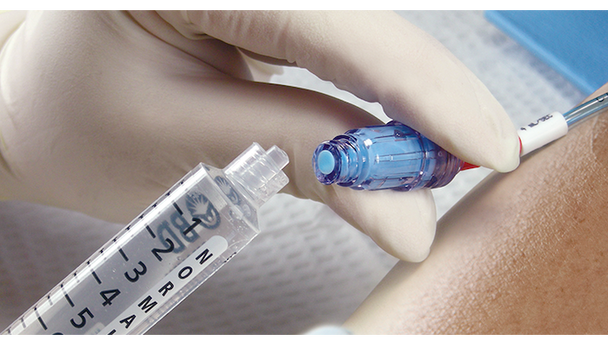
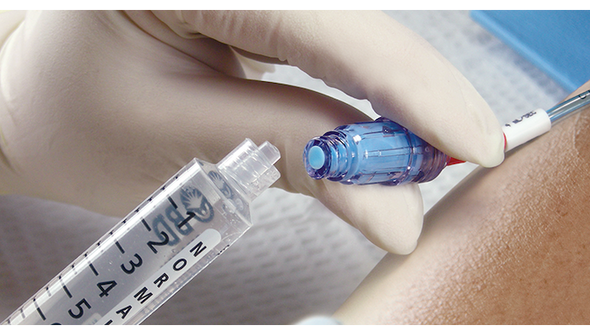
BD Maxplus Needle-Free Connector is designed for clinicians who need reliable, sterile access for IV therapy. This sterile connector helps reduce the risk of contamination and supports efficient workflows in busy hospital and clinical settings. Ideal for healthcare professionals seeking a pressure-rated, needle-free solution that prioritizes safety and ease of use.
- Needle-free Access – Eliminates the need for needles, lowering the risk of needlestick injuries and supporting safer care.
- Sterile and Pressure Rated – Maintains a sterile barrier and withstands pressure, giving clinicians peace of mind during procedures.
- Low Priming Volume – 0.28 mL priming volume helps minimize medication waste, supporting efficient medication delivery.
- Single Connector Design – Features one connector per unit, simplifying setup and reducing complexity for staff.
Reliable Needle-free IV Connector for Clinical Safety and Efficiency
This sterile, pressure-rated connector is engineered for safe, needle-free IV access in hospitals and clinics. The device supports infection control protocols by eliminating needles and maintaining a closed system. Its low priming volume of 0.28 mL helps reduce medication waste, making it a practical choice for high-volume settings. Registered with Health Canada and free from natural latex, this connector is suitable for a wide range of patients. Designed for busy clinical teams, it streamlines IV setup and supports consistent, reliable performance. Choosing this sterile connector means clinicians can focus on patient care with added confidence and efficiency.
Features and Benefits
Clinical Safety and Compliance
- Content Sterile – Ensures a sterile pathway for IV therapy, supporting infection control.
- Pressure Rated – Withstands clinical pressures for safe and reliable use in various procedures.
- Natural Latex-Free – Reduces the risk of allergic reactions for sensitive patients.
- Registered with Health Canada – Meets regulatory standards for clinical use in Canada.
Efficient Medication Delivery
- Low Priming Volume – 0.28 mL volume minimizes medication loss and supports accurate dosing.
- Single Connector Per Unit – Simplifies IV line setup and reduces the chance of confusion or error.
Design and Construction
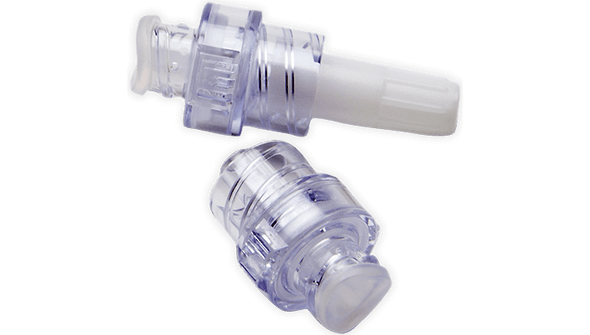
- Needle-free Mechanism – Supports a closed system to help prevent contamination and enhance safety.
- Compact Case Dimensions – 6.25 in × 6.25 in × 5.5 in (15.9 cm × 15.9 cm × 14 cm) for easy storage and handling.
- Country of Manufacture – Made in Mexico for consistent quality.
Product Specifications
Dimensions and Packaging
- Case Dimensions: 6.25 in × 6.25 in × 5.5 in (15.9 cm × 15.9 cm × 14 cm)
- Weight: 1.1 lb (0.5 kg)
- Quantity Per Case: 100
Performance and Safety
- Approximate Priming Volume: 0.28 mL
- Pressure Rated: Yes
- Sterility: Content sterile
- Natural Latex: No
- Registered with Health Canada: Yes
Technical Details
- Number of Connectors: 1
- Type of Connector: MaxPlus
- Country of Manufacture: Mexico
- CE Marking: No
Explore More: See all Needle-free Connectors | Shop more MaxPlus Products
Choose a Needle-free Connector That Supports Safety and Efficiency
When you select this sterile, pressure-rated connector, you are choosing a device that supports infection control, reduces medication waste, and simplifies IV setup. Its low priming volume and needle-free design help clinicians deliver care with confidence and efficiency. This connector is a practical choice for busy hospitals and clinics that demand reliable performance and regulatory compliance.
- Needle-free Safety – Eliminates needles to lower risk of injury and contamination, supporting a safer environment.
- Low Medication Waste – 0.28 mL priming volume helps conserve medication, which is important for cost control.
- Single Connector Simplicity – One connector per unit streamlines setup and reduces the chance of error.
- Latex-Free Construction – Suitable for patients with latex sensitivities, offering peace of mind.
- Regulatory Compliance – Registered with Health Canada, meeting important clinical standards.
- Pressure Rated Advantage – Designed to withstand clinical pressures, providing a measurable edge over non-rated connectors.
Frequently Asked Questions (FAQs)
What is the priming volume of this connector?
The priming volume is approximately 0.28 mL, which helps minimize medication waste.
Is this connector sterile?
Yes, the connector is supplied in a sterile condition to support infection control protocols.
Does this product contain natural latex?
No, this connector is free from natural latex, making it suitable for sensitive patients.
How many connectors are included per case?
Each case contains 100 connectors for efficient supply management.
Is this connector pressure rated?
Yes, it is pressure rated for safe use in various clinical procedures.
Where is this connector manufactured?
This connector is manufactured in Mexico, ensuring consistent quality.
Is the connector registered with Health Canada?
Yes, it is registered with Health Canada for clinical use in Canada.