Description
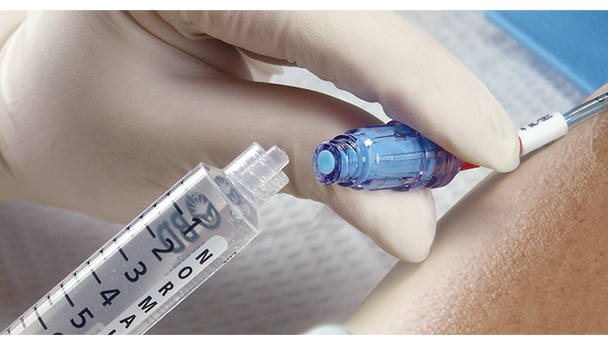
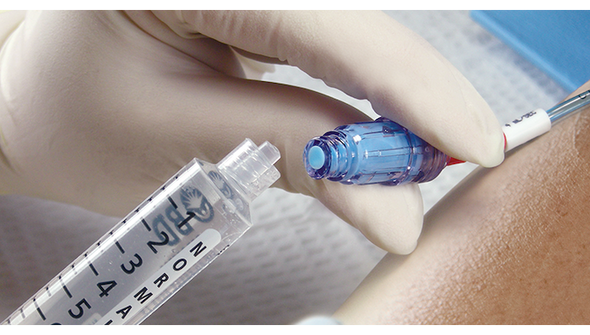
BD Maxplus Clear Needle-Free Connector is designed for clinicians who need reliable, sterile access for IV therapy. This connector supports safe, efficient workflows and helps reduce the risk of contamination. Ideal for hospital and clinical settings where pressure-rated, needle-free access is essential for patient safety and staff confidence.
- Pressure Rated – Handles high-pressure infusions, giving you peace of mind during critical procedures.
- Needle-free Access – Reduces the risk of needlestick injuries, supporting a safer environment for staff.
- Sterile Content – Ensures each connector is ready for immediate use, helping you maintain infection control.
- Clear Housing – Allows easy visual inspection, so you can quickly confirm flow and cleanliness.
- Low Priming Volume – 0.28 mL priming volume minimizes medication waste and supports precise dosing.
Reliable Needle-free IV Connector for Safe, Efficient Infusion
This sterile, pressure-rated connector is engineered for safe IV access without needles, supporting infection control and staff safety. The clear housing lets you visually verify flow and cleanliness, while the low 0.28 mL priming volume helps reduce medication waste. Designed for busy clinical environments, this device supports efficient workflows and helps lower the risk of contamination. Hospitals and clinics that require reliable, needle-free connectors for IV therapy will appreciate the safety and efficiency this product brings to daily routines.
Features and Benefits
Safety and Infection Control
- Needle-free Design – Eliminates the need for needles, reducing exposure risk for staff and patients.
- Sterile Content – Each unit is supplied sterile to support infection prevention protocols.
Performance and Efficiency
- Pressure Rated – Suitable for high-pressure infusions, supporting a wide range of therapies.
- Low Priming Volume – 0.28 mL volume helps minimize medication loss and ensures accurate dosing.
Visibility and Ease of Use
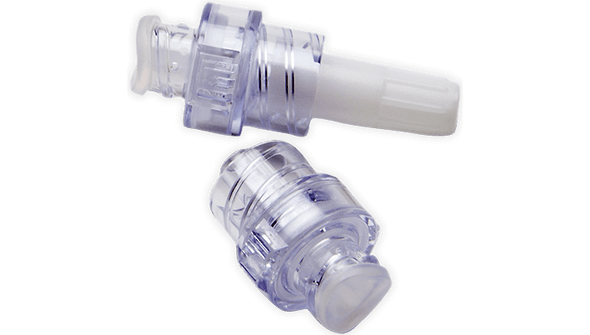
- Clear Housing – Lets you easily inspect for flow and cleanliness at a glance.
- Single Connector – Simple, one-connector design streamlines setup and use.
Product Specifications
Dimensions and Weight
- Case Dimensions: 6.25 in x 6.25 in x 5.5 in (15.9 cm x 15.9 cm x 14 cm)
- Weight: 1.1 lb (0.5 kg)
Technical Details
- Approximate Priming Volume: 0.28 mL
- Pressure Rated: Yes
- Number of Connectors: 1
- Sterility: Content sterile
- Type of Connector: MaxPlus clear
- Natural Latex: No
- Registered with Health Canada: Yes
- Country of Manufacture: Mexico
Packaging
- Quantity Per Case: 100
- Unit of Measure: Per case
Explore More: See all Needle-free Connectors | Shop more MaxPlus Products
Choose Reliable, Sterile Needle-free Access for Your Facility
When you need a connector that supports infection control and workflow efficiency, this device stands out for its sterile, pressure-rated design. The clear housing and low priming volume help you deliver care with confidence. Compared to generic connectors, this product is engineered for safety and reliability in demanding clinical environments.
- Pressure-rated Performance – Handles demanding infusions, supporting a wide range of therapies with confidence.
- Sterile and Latex-free – Reduces allergy risk and supports infection prevention protocols.
- Clear Visual Inspection – Transparent housing lets you confirm cleanliness and flow instantly.
- Low Medication Waste – 0.28 mL priming volume helps minimize loss and supports precise dosing.
- Registered with Health Canada – Meets regulatory standards for safety and quality.
Frequently Asked Questions (FAQs)
What is the priming volume of this connector?
The priming volume is approximately 0.28 mL, which helps reduce medication waste.
Is this connector pressure rated?
Yes, this connector is pressure rated for high-pressure infusions.
Does the product contain natural latex?
No, this connector is latex-free to reduce allergy risk.
How many connectors come in a case?
Each case contains 100 connectors.
Is the connector supplied sterile?
Yes, each connector is supplied sterile for infection control.
Where is this connector manufactured?
This product is manufactured in Mexico.
Is this connector registered with Health Canada?
Yes, it is registered with Health Canada for regulatory compliance.